Local Anesthetics have been one of the most important inventions in human history. You may recognize many of these terms in this article if you took a high school chemistry class. Please visit our glossary at the end of this article to review/learn them.
Many of our patients at our Mississauga dental office ask us how they work and are interested in their chemistry. Local anesthetics are the most frequently used drugs at St. Lawrence Dentistry, and we administer thousands of doses each year. These are drugs used to relieve pain in your mouth without the loss of consciousness, and they do this by blocking nerve conduction. More specifically, they block “sodium channels”, which block a nerve transmission or “action potential”. Consequently, there is no message sent to the brain.
Dr. Hawryluk Jr. thinks the cell biology of local anesthetics is fascinating. A natural charge difference usually exists between the inside and outside of a cell. When a nerve “depolarizes”, it opens a “sodium channel”, which allows a nerve action potential to occur. Local anesthetic molecules sit in the “open state” of a channel and prevent sodium passage. Consequently, you will feel no pain during a dental procedure with local anesthetics.
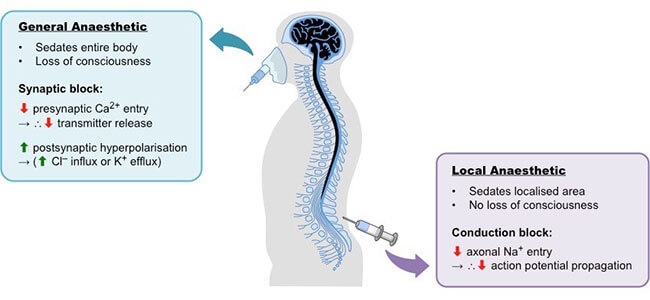
St. Lawrence Dentistry pays close attention to the dosages and duration of local anesthetics. An increased dose of a local anesthetic means more molecules will arrive at the injection area. The “onset” of the anesthetic may be quicker with an increased amount. Important factors which determine how effective a local anesthetic will be are the pH of the tissue and the pKA of the local anesthetic. pKa is the relative concentration of the local anesthetic in the “nonionized lipid-soluble” form versus the “ionized water-soluble” form. The ionized form of local anesthetics is the one that can penetrate a cell’s membrane, and this is what makes it effective. However, the nonionized form is not as effective since it cannot penetrate cell membranes.
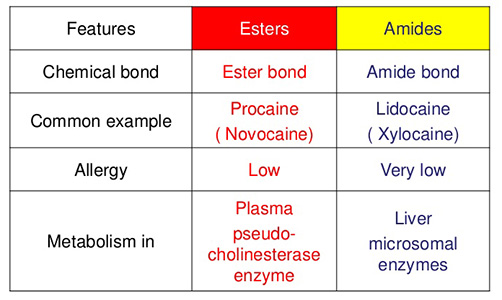
There are two categories of modern local anesthetics: amides and esters. The amide locals are the most commonly used at St. Lawrence Dentistry and have pKa ranging from 7.9-7.6. The other type is the Esther locals are who are closer to pKa 9. The closer the pKa is to the tissue pH, the more ionized form available and the more effective it will be.
Understanding this principle becomes important when treating dental infections as the pH of the infection can be more acidic. Local anesthetics closest to the physiological pH will have a higher concentration of nonionized base form. Therefore it can more easily pass through nerve cell membranes to give a quicker onset of action. St. Lawrence Dentistry will consider the probable pH of your tissue when selecting the most appropriate anesthetic for you. For example, if injecting the common local lidocaine (pKa of 7.9) into an infected area with a pH of 5.9 will only allow a 1000:1 ionized versus nonionized form – this means only a fraction is available for diffusion into a cell. All in all, you would find it would not work well- this is why Dr. Hawryluk sometimes prescribes antibiotics first waits for an infection to clear before doing a dental procedure.

Some patients ask us what the components of local anesthetic are. A local anesthetic molecule consists of a lipophilic centre (aromatic group), a hydrophilic centre (tertiary amine), and an intermediate group (amide or ester). One interesting fact is Bupivicaineis the longest-lasting amide anesthetic which can last 9 hours.

When using local anesthetics, we are always taking steps to avoid adverse reactions. These can be psychogenic, allergy, toxicity, and paraesthesia-related. Syncope is the most common psychogenic side effect; it is related to an increase or decreases in heart rate and blood pressure. It is usually the result of unintentional epinephrine injection into a vessel. Allergic reactions to local anesthetics are infrequent and generally related to a compound “p-aminobenzoic”. Historically, there were allergies to methylparaben in anesthetics. However, this compound is not in modern solutions. There are no true allergies to epinephrine in anesthetics as this is an “endogenous neurotransmitter” that naturally occurs in your body. Toxicity to local anesthetics is rare, but it can be related to excessive dosages or direct injection into a vessel.
A common question we get at St. Lawrence Dentistry is about how we calculate the maximum dose for local anesthetics. Our most common anesthetic used at our office are lidocaine and articaine, and they both have a maximum dosage of 7mg/kg. When you see a percentage listed on a local anesthetic carpule, this is in the units grams/100ml. Hence 2% lidocaine has 20mg/ml, and each cartridge has 1.8ml. Accordingly, for a 25kg child, the max dose would be calculated as follows.
- Max dose is 7mg/kg
- 7mg x 25kg=175mg
- 2% lido has 20mg/ml
- 175mg/ 20mg/ml=8.75ml
- 8.75ml/1.8ml=4.86 cartridges

One rare complication of local anesthetics is parasthesia. It is a partial loss of sensation and associated with a burning, tingling feeling and usually resolves in 4-6 weeks. Local anesthetics containing 4% solutions have a higher percentage of paresthesia. Hence, at St. Lawrence Dentistry, we mainly use a 2% solution.
Hematomas are another rare complication for anesthetics and result from leakage of blood from vessels into the tissue. Statistics have shown that the most likely instance for this to happen is after the dentist gives a “PSA nerve block” – an injection to numb your upper teeth. These usually heal with discoloration, and most of the time, no further treatment is needed.
If a patient has a hematoma, we generally will advise the following: Apply ice for the next 6 hours 15 mins on 15 off. Avoid aspirin or and non-steroidal anti-inflammatory medications as they may cause bleeding.

Trismus is another rare anesthetic complication and is a motor disturbance of the trigeminal nerve characterized by the inability to open the mouth. The muscle mostly affected is called the “middle pterygoid”.
If you have trismus, the treatment we sometimes recommend will be: Place hot moist towels 20mins on/10min off, use analgesics, and practice opening and closing. In this instance, we have found ibuprofen 600mg strength is helpful.

Epinephrine is contained in many local anesthetics and decreases the local absorption rate into the cardiovascular system. Hence less chance of toxicity. It also reduces bleeding, increases the duration of action of the local. Epinephrine does react with medications, especially beta-blockers, tricyclic antidepressants, and amphetamines. Generally, this will manifest as an increase in blood pressure and heart rate.
If we have any concerns about this happening, we will lower the dose of epinephrine used. We will generally use below 2 “carpules” of 1:200k epinephrine for our patients with cardiovascular issues.

St. Lawrence Dentistry will do everything possible to make your dental appointment comfortable with the help of local anesthetics. If you have any questions about their interesting chemistry please let us know.
References: University of Toronto, Faculty of Dentistry online Lecture series 2018.
Glossary of Terms
- Nerve: a fiber in the body which transmits impulses of sensation to the brain.
- Action potential: the change in electrical potential associated with the passage of a nerve impulse.
- Sodium channel: a channel in a cell that allows electrical signals to pass through.
- Cell: the smallest unit of a living being.
- Depolarization: a shift in electrical charge distribution within a cell.
- pH: a measure of acidity level.
- Ionization: The addition or removal of electrons from an atom.
- Physiological pH: the pH which prevails in the human body
- Endogenous neurotransmitter: chemical messengers which transmit signals from nerve to a cell.
- Syncope: the temporary loss of consciousness caused by a fall in blood pressure.
- Epinephrine: the drug form of the hormone adrenaline (which the body produces on its own).
- pKa: a value that indicates how well an acid will dissolve in water.
- Understanding Dental Cupping on Molars: Causes, Prevention, and Treatment - August 14, 2024
- Bone Preservation: Essential for Strong Dental Implants - August 5, 2024
- Porcelain Veneers from Design to Finish - July 27, 2024