St. Lawrence Dentistry is pleased to offer the Mississauga community a new groundbreaking dental material. It’s called GC fuji auto mix LC, and its classification is a Resin reinforced glass ionomer.
Made by the renowned dental material developer, GC America, Its indicated for children’s dental care (pediatric dentistry) and has many uses in adults.

This article contains phrases and terms which are common in chemistry study. For more enjoyable reading, please visit our glossary at the end of this article. Of course, feel free to discuss them with Dr. Hawryluk Jr. when you are at our Mississauga Dental Clinic.

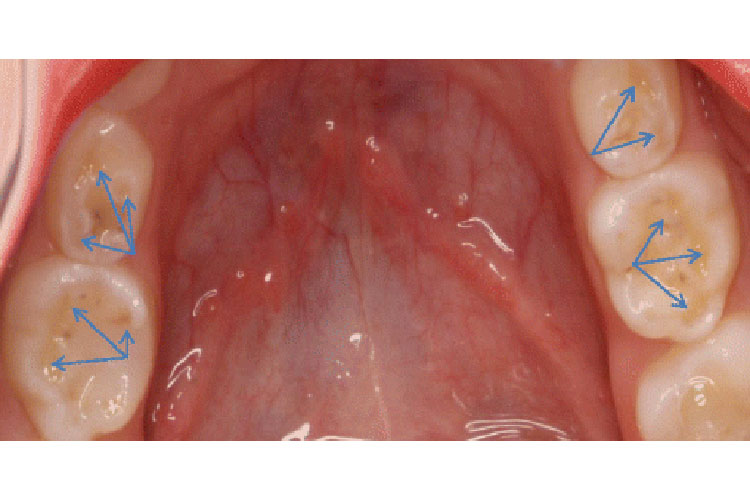
A glass ionomer is one of the main classes of dental filling materials that are unique in that it can release fluoride and bond to a tooth. Not only is this beneficial to children’s dentistry, but it also has adult applications. GC fuji auto mix LC is a special glass ionomer with several benefits. The small particle size in the Fuji GC auto mix allows for excellent polishability and aesthetics. Some materials feel rough or grainy and do not feel completely smooth— St. Lawrence Dentistry does have this issue with the Fuji material. Most glass ionomers are in a ‘capsule-based system.’ This product is in a syringe system, making the whole process of fixing your tooth less time consuming and exceptionally accurate. The delivery system helps Dr. Hawryluk adapt the material into all the little nooks and crannies in a tooth without leaving any voids. These benefit mean shorter appointment times for children dentistry (pediatric dentistry), which is essential as children often don’t like to sit for too long. The material efficiencies also mean less general anesthetic time when Dr. Hawryluk is performing dentistry asleep, in conjunction with our Board Certified Medical Anesthesiologists.

Residents of Mississauga expect and want the most modern dental materials. The number one uses for this material is underneath a sizeable composite restoration near to the dental pulp. Flowing Fuji GC here will encourage reparative dentin formation and can prevent your tooth from needing a root canal. No acidic acid etch (like dental composite material) is required, and so the tooth is not exposed to this harsh conditioner making it less likely for internal tooth injury. The other prime use is to fill in hard to reach nooks and crannies and under dental crowns. Resin-modified glass ionomers allow equal or higher fluoride release (compared to regular glass ionomers). There is evidence of higher retention to a tooth, higher strength, and lower solubility in the mouth. The fluoride in this product can recharge from being exposed to fluoride in toothpaste therefore, it is a biodynamic or bioactive material. Recharging the materials fluoride concentration has been shown to reduce cavity risk. It can provide subsurface remineralization under a dental restoration.

Patients often ask Dr. Hawryluk Jr. about the release of fluoride is from Glass ionomers. The fluoride release pattern has an initial rapid release of considerable amounts of fluoride, followed by a taper in the release rate over time. An initial fluoride burst effect is desirable to reduce the viability of remaining bacteria in the inner carious dentin, hence, inducing enamel or dentin remineralization. The constant fluoride release during the following days is attributable to the fluoride ability to diffuse through cement pores and fractures. Thus, continuous small amounts of fluoride surrounding the teeth reduce the demineralization of the tooth tissues.
The chemistry of the material setting is taught right at our local high schools — such as Port Credit and Lorne Park Secondary School— where Dr. Hawryluk attended. Resin-based glass ionomers have two setting reactions: an acid-base setting and a free radical polymerization. The chemical bond (acid-base setting) of the glass ionomer to the tooth is by an ionic exchange bonding process. A strong chemical bond is made by polyacrylic acid, displacing some of the ions under the tooth surface. There is a secondary bond of the material bonding to collagen within the dentin matrix. Activation of the free-radical polymerization is by light and is the predominant mode of setting, as it occurs more rapidly than the acid-base setting which is comparatively slower.

The Resin bonded glass ionomer has some similarities to dental composite material. The strength of Resin reinforced glass ionomers rivals that dental composite but has the added benefit in that it chemically fuses with the tooth and release fluoride. When you consider that composite materials have the risk of leakage and shrinkage, glass ionomers have overwhelming advantages.

In addition to filling teeth with glass ionomers, they also serve in cementing dental crowns. Dr. Hawryluk still uses Resin cement (instead of glass ionomer) to lute if a using All-ceramic lithium disilicate crowns (Emax Crowns). However, if cementing Zirconia Crowns, or porcelain fused to metal crowns, he is always reaching for the glass ionomer due to the cavity resistance and the bond to the tooth, and excellent seal it provides.
It is crucial to realize the design and engineering of your dentistry by Dr. Hawryluk is far more critical than the type of material used. Nothing will make up for a poorly designed restoration or lack of ferrule in tooth design. St. Lawrence Dentistry always adheres to the fundamentals of excellent preparation design for your dental work. Our goal is to provide the best Mississauga Dental Care. There is proof that the Fuji GC auto-mix has a strong bond to the tooth structure and other restorative materials. Excellence in bonded dentistry and a happy patient is our ultimate goals.
Glossary of Terms:
- Ion – an atom or molecule with an electric charge due to the loss or gain of one or more electrons.
- Atom – the smallest constituent of ordinary matter that constitutes a chemical element.
- Ionic – a chemical bond formed by the electrostatic attraction of oppositely charged ions.
- Ionomer – any class of polymer materials consisting of thermoplastic resins stabilized by ionic cross-linkages.
- Lithium disilicate crown – a dental crown composed of quartz, lithium dioxide, phosphor oxide, alumina, potassium oxide, and other components. Overall, this composition yields a great thermal shock resistant glass-ceramic due to the low thermal expansion that results when it is processed.
- Solubility – the ability to be dissolved, especially in water.
- Fluoride – a fluorine(a chemical element) containing salt which can reduce tooth decay.
- Free radical – an uncharged molecule (typically highly reactive and short-lived) having an unpaired valence electron.
- Electron – a stable subatomic particle with a charge of negative electricity, found in all atoms and acting as the primary carrier of electricity in solids.
- Valence – a whole number which represents the ability of an atom or a group of atoms to combine with other atoms or groups of atoms. Determining the valence is by the number of electrons that an atom can lose, add, or share.
- Free-radical polymerization – is a polymerization method by which polymer forms by the successive addition free-radical building blocks.
- Glass ionomer cement – is a class of dental restorative materials used by St. Lawrence Dentistry. The basis of their chemistry is on the reaction of silicate glass powder calcium-alumino-fluorosilicate glass and polyacrylic acid, an ionomer. This reaction produces a powdered cement of glass particles surrounded by a matrix fluoride and is known chemically as glass polyalkenoate.
- Carious dentin – tooth decay in the dentin layer of a tooth.
- Polyacrylic acid – is a synthetic polymer.
Reference: P.Maragliano: Viva learning: “innovations in Resin modified glass ionomers, 2020
- Understanding Dental Cupping on Molars: Causes, Prevention, and Treatment - August 14, 2024
- Bone Preservation: Essential for Strong Dental Implants - August 5, 2024
- Porcelain Veneers from Design to Finish - July 27, 2024